Monitoring substance use from home has become a practical reality for people in recovery. At EveningIOP, we recognize that remote drug testing options offer flexibility without compromising accountability.
This guide walks you through how at-home testing works, compares it to traditional methods, and helps you find the right solution for your situation.
How At-Home Drug Testing Actually Works
Urine and Saliva Testing Methods
Urine-based testing remains the most practical option for remote monitoring because the collection process is straightforward and the accuracy rivals laboratory results when performed correctly. You collect a sample in a cup, apply it to a test device, and read results within minutes-typically detecting marijuana, cocaine, opiates, methamphetamine, amphetamines, PCP, benzodiazepines, barbiturates, methadone, tricyclic antidepressants, MDMA, and oxycodone. Saliva-based tests offer a faster alternative, delivering results in as little as 10 minutes with minimal discomfort, though they work best for substances with shorter detection windows.
Detection Windows and Timing
The key difference between these methods lies in specimen stability and drug clearance rates. Detection windows for different substances in drug testing vary by substance and individual factors. Testing too early after suspected use can produce false negatives, while testing weeks later may also miss recent consumption depending on the substance.
Sample Validity Checks
At-home kits include built-in safeguards to catch tampering or degraded samples. Temperature strips verify the urine is fresh, while chemical checks measure creatinine levels, pH, specific gravity, nitrites, and oxidants to confirm the sample hasn’t been diluted or contaminated. A telehealth-based opioid treatment program studied by Williams et al. in JAMA Health Forum (2023) administered 16-panel kits to 3,395 patients, with 81.9% completing urine drug testing within 30 days and 99.7% within 180 days, demonstrating that remote testing is both feasible and scalable.
The number 100% seems to be not appropriate for this chart. Please use a different chart type.
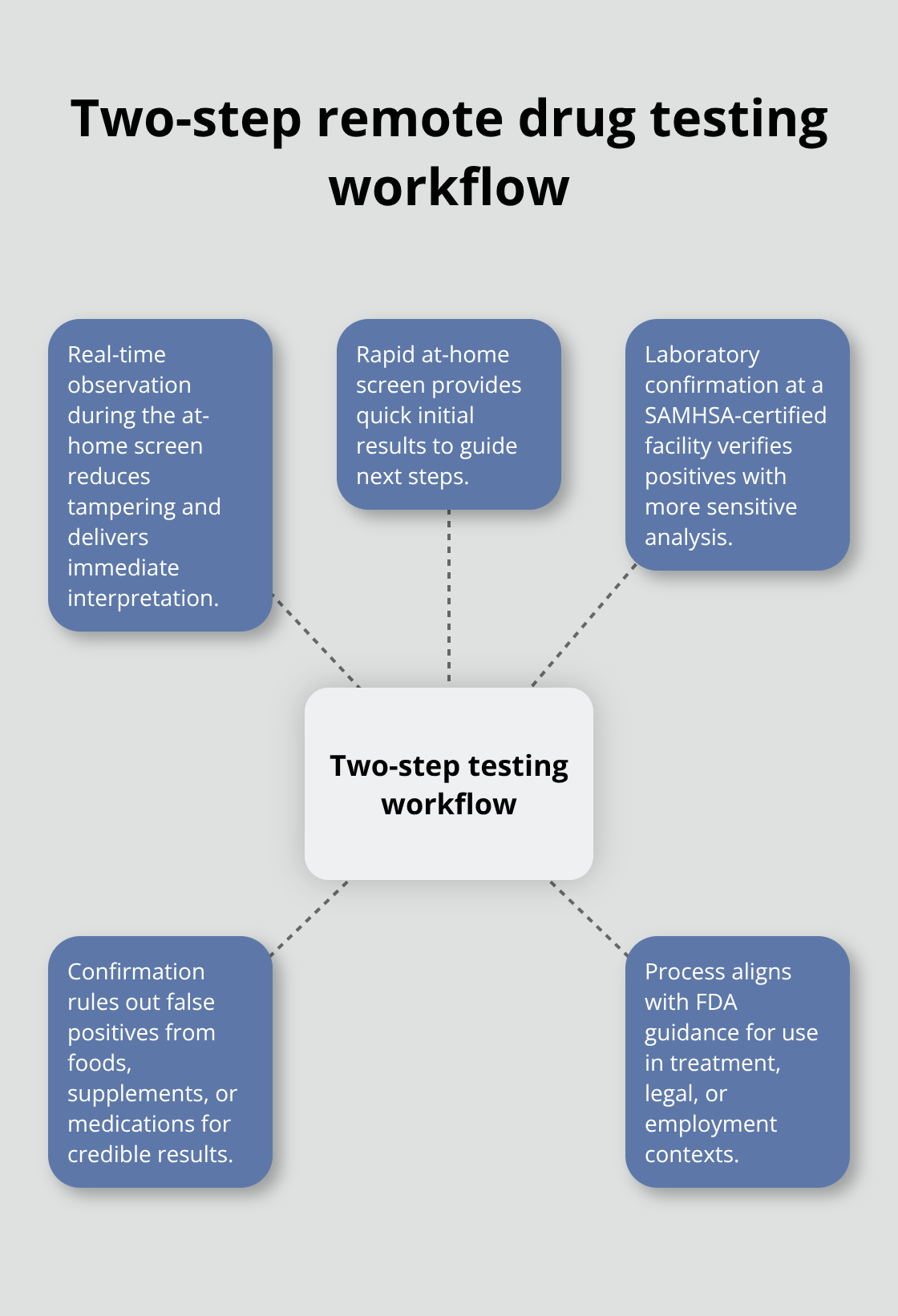
Real-Time Verification and Lab Confirmation
The study used real-time visual interpretation during telehealth visits, meaning a clinician watched the test being performed and reviewed results immediately, which significantly reduced tampering risk. When a positive result appears, the sample travels to a SAMHSA-certified laboratory confirmation testing facility, which uses more sensitive chemical analysis to verify the presence of drugs and rule out false positives from foods, supplements, or medications. This two-step approach-rapid at-home screening followed by laboratory confirmation-aligns with FDA guidance and provides credible evidence suitable for treatment monitoring, legal proceedings, or employment verification.
Cost Advantages of Remote Testing
Home testing bypasses third-party lab facility fees, lowering expenses for employers and individuals while maintaining reliability through proper specimen handling and confirmation protocols. These cost savings, combined with the ability to test from your own space, make virtual therapy and remote monitoring accessible for people managing recovery alongside work and family commitments. The next section compares how remote testing stacks up against traditional in-person approaches, helping you understand which method fits your specific needs and circumstances.
Remote and In-Person Testing: What Actually Differs
Accuracy Holds Steady Across Both Methods
Remote testing produces results as reliable as in-person lab work when specimens are properly collected and verified. The Williams et al. study published in JAMA Health Forum tracked 3,395 patients in a telehealth opioid treatment program and found that 81.9% completed urine drug testing within 30 days using at-home kits, with 99.7% completing tests within 180 days. Real-time visual observation during telehealth visits supports treatment adherence monitoring. The study used specimen validity checks including temperature, creatinine, pH, specific gravity, and chemical markers to confirm sample integrity. When positive results appeared, samples went to SAMHSA-certified laboratories for confirmation using more sensitive chemical analysis, matching the gold standard accuracy of traditional lab-only workflows.
Where Verification Happens Matters Most
The key difference isn’t accuracy-it’s location and timing. In-person testing requires you to travel to a collection site, wait in a facility, and follow that location’s scheduling. Remote testing lets you collect at home on your timeline, then the same laboratory confirmation process verifies the sample. For people juggling work schedules or managing recovery alongside family responsibilities, this flexibility removes a significant barrier to consistent monitoring.
Cost Savings Add Up Over Time
Traditional in-person testing involves facility overhead, staff time, and logistics costs that get passed to employers or individuals-sometimes $50 to $100 per test. At-home kits bypass these middleman expenses, reducing costs substantially while maintaining identical confirmation standards. The real advantage appears over time. If you need monthly monitoring for six months, the cumulative savings become substantial, especially when you factor in the time cost of traveling to collection sites.
When In-Person Testing Still Makes Sense
In-person testing remains valuable for initial screening or legal proceedings where chain-of-custody documentation must be airtight and observed from start to finish. For ongoing progress verification in treatment, however, remote testing with laboratory confirmation provides credible evidence at a fraction of the cost. This distinction shapes how treatment programs structure their monitoring approach-and it directly influences which testing method fits your recovery plan.
Choosing the Right Remote Testing Provider
Verify Laboratory Certification and Specimen Safeguards
Select a remote testing provider by focusing on three concrete factors: specimen validity safeguards, laboratory certification, and integration with your treatment program. The provider you choose must use SAMHSA-certified laboratories for confirmation testing-this is non-negotiable if results need to hold up in legal or employment contexts. Beyond certification, verify that their at-home kits include temperature strips and chemical validity checks for creatinine, pH, specific gravity, nitrites, and oxidants. The Williams et al. study in JAMA Health Forum tracked 3,395 patients using 16-panel kits with these safeguards built in, and 81.9% completed testing within 30 days, demonstrating that proper specimen validation directly supports consistent monitoring.

Prioritize Real-Time Observation During Collection
Ask potential providers whether they offer real-time visual interpretation during collection-a clinician watching the test happen eliminates tampering concerns and accelerates result verification. Some providers mail kits without observation, which cuts costs but increases tampering risk; others charge premium rates for synchronous administration. The middle ground works best: kits mailed to you with the option for real-time observation during telehealth visits, combining affordability with accountability.
Align Testing with Your Treatment Program’s Workflow
Integration with your treatment program matters more than most people realize. If you’re in an IOP program, the testing provider must connect seamlessly with your clinician’s workflow-results should appear in a shared portal within hours, not days, so your treatment team can adjust your care plan based on actual data. Ask whether the provider offers automated reporting to your program, secure result portals for you and your clinician, and whether they support the specific drug panels your treatment requires. Some providers offer only standard 5-panel or 10-panel tests, while others customize panels to exclude THC or include medications specific to your situation.
Evaluate Pricing and Report Quality
Pricing varies widely: home kits range from $15 to $50 per test when purchased individually, but bulk agreements with treatment programs often drop costs to $10 to $20 per test. Request a sample report before committing-it should clearly distinguish between preliminary at-home results and confirmed laboratory results, use language your clinician understands, and flag any validity issues immediately.
Confirm HIPAA Compliance and Data Protection
Privacy protection is straightforward: confirm the provider is HIPAA-compliant, uses encrypted portals for result transmission, and stores data on secure servers. Your results should never be shared with employers or insurers without explicit written consent, and the provider should have a clear policy on result retention and deletion timelines.
Final Thoughts
Remote drug testing options transform recovery by removing barriers that prevent consistent monitoring. The Williams et al. study proved this reality: patients in a telehealth opioid treatment program completed tests at rates exceeding 99.7% over 180 days, with declining illicit opioid positivity demonstrating that remote monitoring supports genuine recovery outcomes when paired with clinical support. What matters most is not the test itself but the pattern over time and how your clinician interprets results within your treatment context.
The strength of remote testing emerges when it integrates into comprehensive care that combines therapy, medication management, and ongoing clinical guidance. We at EveningIOP embed remote drug testing directly into our telehealth evening Intensive Outpatient Programs, meaning your test results inform your treatment plan in real time and adjustments follow immediately. This approach lets you pursue recovery without sacrificing work or family commitments.
Start by identifying a provider whose laboratory certification and specimen safeguards align with your treatment needs. Explore how EveningIOP structures remote monitoring within comprehensive evening treatment to see whether this model fits your recovery strategy. The right remote testing solution removes friction from recovery without compromising accountability.


